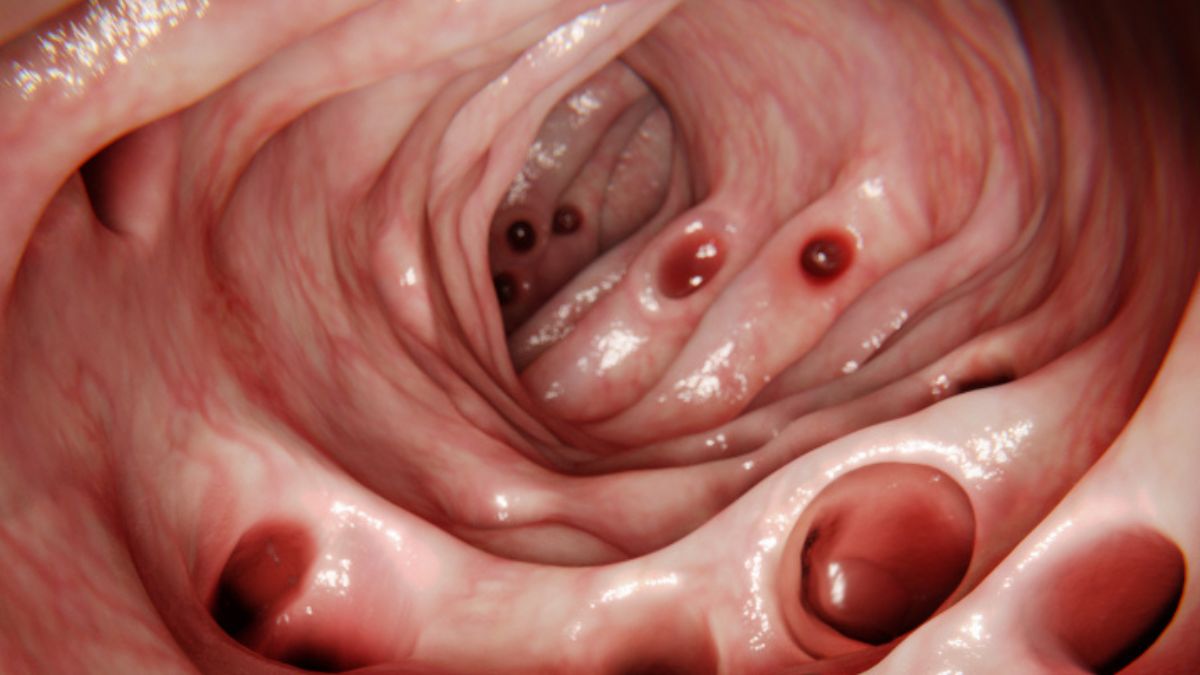
A miniature model of the human uterus just menstruated in a petri dish for the first time. Published today in Nature, this breakthrough reveals how endometrial tissue sheds and repairs itself without scarring—a process that could transform treatments for infertility, endometriosis, and even wound healing.
The Science

Researchers at the Institute of Molecular Biology in Vienna engineered endometrial organoids: 3D structures derived from stem cells that mimic the uterine lining. By exposing them to a simulated 28-day hormonal cycle, the organoids responded with genetic and structural changes identical to the human endometrium—proliferation, differentiation, and ultimately, menstrual shedding. For the first time, a lab model captures the full cycle. The team used cells from healthy donors and cultured them in a collagen matrix, mimicking the stiffness of uterine tissue. During the luteal phase, the organoids secreted vascular endothelial growth factor (VEGF), promoting angiogenesis necessary for regeneration. This process was followed by activation of caspases, the enzymes that execute apoptosis, allowing controlled cell removal without uncontrolled inflammation.

The key finding is that endometrial stromal cells, when exposed to progesterone, activate a Wnt signaling pathway that triggers a controlled inflammatory response. This inflammation is necessary for tissue breakdown, but unlike other wounds, repair factors like matrix metalloproteinase 9 (MMP-9) and bone morphogenetic protein 7 (BMP-7) are simultaneously activated. The result: tissue degrades and rebuilds without fibrosis. In mice with endometrial injuries, applying BMP-7 reduced scar formation by 40%. The researchers also observed that the Wnt pathway is rapidly deactivated after menstruation, preventing excessive proliferation that could lead to cancer. This temporal balance is crucial: in pathological conditions like endometriosis, the Wnt pathway remains active, suggesting that dysregulation of this mechanism could be an underlying cause.