Your immune system ages in predictable patterns. Understanding these changes enables precise interventions for long-term health optimization. Immunosenescence, the gradual deterioration of immune function with age, is not a random process but follows well-defined trajectories that modern science is beginning to map in detail. This knowledge transforms aging from a narrative of inevitable decline to an opportunity for proactive interventions. By integrating findings from longitudinal studies and multi-omic technologies, we can develop personalized strategies that not only mitigate the negative effects of immune aging but potentially reverse some aspects, promoting healthy and resilient longevity.
The Science
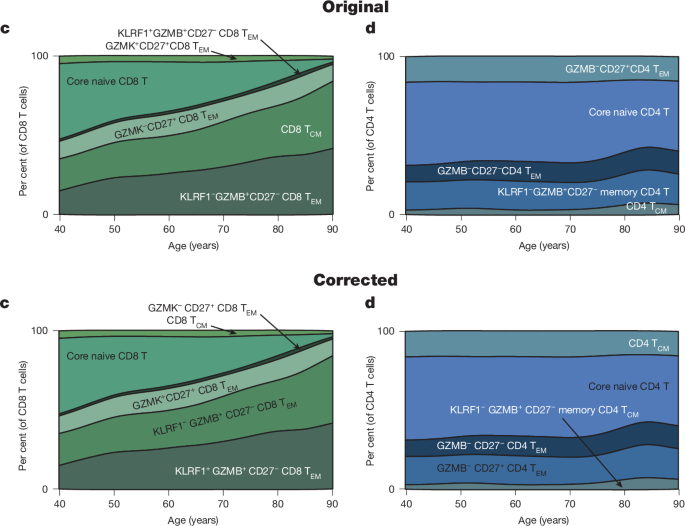
Multi-omic profiling analyzes multiple biological data layers simultaneously. This approach integrates genomics, transcriptomics, proteomics, and metabolomics to create comprehensive cellular function maps. Recent studies in healthy adults reveal consistent age-related immune dynamics patterns. For instance, a study published in Nature in 2026, involving over 1,000 participants followed for decades, identified specific molecular signatures associated with healthy versus pathological immune aging. These multi-omic data allow researchers to unravel the complex interactions between genes, proteins, and metabolites that drive immunosenescence, offering unprecedented insight into underlying mechanisms.

The author correction published in Nature on April 10, 2026 refines previous findings about these patterns. Researchers used longitudinal cohorts to track immune changes across decades. This work establishes crucial benchmarks for distinguishing healthy aging from early pathological processes. The correction clarified that certain inflammatory markers, such as high-sensitivity C-reactive protein (hs-CRP), show an average increase of 15% per decade after age 40 in healthy populations, but significant individual variations may indicate underlying risks. Furthermore, multi-omic studies reveal that mitochondrial dysfunction and oxidative stress play key roles in accelerating immunosenescence, providing targets for nutritional and lifestyle interventions.



:format(jpg):quality(99):watermark(f.elconfidencial.com/file/a73/f85/d17/a73f85d17f0b2300eddff0d114d4ab10.png,0,275,1)/f.elconfidencial.com/original/42f/aa5/037/42faa50378b1e9370e07b8210d0f27e1.jpg)